Safety is a very important aspect of laboratory activity. Without it, daily undertakings can lead to injuries, severe health risks, and even death. To reduce these risks, there are certain procedures, also called Good Laboratory Practices (GLP), that we should all follow in the laboratory as individuals and as a member of a group.
When it comes to safety related to chemical handlings, it is crucial to protect yourself from toxic fumes and other vapors, hence avoiding any inhalation of chemicals. This safety can only be accomplished with the systematic usage of laboratory fume hoods which is the only equipment nowadays guaranteeing user’s protection from inhalation of chemicals. Different types of fume hoods are currently available as we can adopt a traditional ducted system (featuring exhaust pipe) or a ductless system (featuring filtration technology). Regardless of the category of fume hood, all units should comply with the safety requirements given by International Safety Standards in Europe, North America and Asia. Unfortunately, many fume hoods available in the market do not meet these safety requirements mainly due to manufacturer’s poor expertise about fume hood, due to the lack of test and certification before and after installation, and due to lack of understanding, on how to use a fume hood safely, by the chemists.
Therefore, it is fundamental for any lab manager and lab personnel to differentiate between good and bad fume hoods, or again between safe fume hoods and risky fume hoods, and to discipline chemists to use the fume hoods properly. Safety awareness in the laboratory starts with familiarizing yourself with all equipment and proper procedures before using your fume hood. Throughout this article, you’ll read much on the importance of laboratory fume hood and the related safety requirements that it must comply with. Basically it will reply to the question: how do I know if my fume hood (or my future fume hood) is considered safe or not?
Safety and chemical exposure
Before going more into technical details with fume hoods, it’s necessary to have a clear understanding about the current safety regulations set in place for chemical exposure. To put it simply, what are the average chemical concentrations to which workers can be exposed at workplace?
Inhaling chemical presents potential risks for your health after a certain level of exposure. It can lead to health consequences on specific or several organs, weaken the immune system, affect the blood and bone marrow or even cause death. These consequences can be short and long term, depending upon the severity of chemical exposure.
In USA, laboratory personnel follow strict guidelines from the National Institute for Occupational Safety and Health (NIOSH), while similar regulations in Malaysia are given by the Department of Occupational Safety and Health (DOSH), under the occupational safety and health act, for use and standards of exposure of chemicals hazardous to health.
In order to ensure end-user’s safety, these organizations have determined a regulatory limit on the amount or concentration of a substance in the air. These limits, or allowable airborne concentrations, are known under the designation Permissible Exposure Limit (PEL). We can also find them under the names Threshold Limit Value (TLV) and Occupational Exposure Limit (OEL) depending on the said country.
On a safety matter, a person inhaling concentrations higher than the PEL-TWA of chemicals at workplace regularly will imply a health risk. Indeed, the daily exposure to low volume of chemicals may not have immediate health consequences but it does on the long term. Hence, it is crucial for all individuals to minimize their chemical exposure at a threshold value much below the PEL-TWAs, since PEL-TWA values are determined for one chemical only. It does not take in consideration the possible accumulation with other chemicals present in the same room, or even the potential mix of different chemicals, which is the case in a chemistry lab.
A chemistry laboratory is a unique working environment with the presence of many toxic products in the room, resulting in the possible exposure to multiple chemicals at the same time. Therefore, there is an accumulation of different concentration of chemicals in the laboratory, that could be inhaled by the worker. Besides, there is also the risk of a combination of different chemicals, also named “cocktail effect”, which may result in a very dangerous mix. This mix may represent a much higher danger in terms of toxicity and be insidiously inhaled by the user. Eventually, the accumulation and mixture of chemicals can have dramatic health consequences for a chemist despite his respect for PELs-TWA values.
That’s the reason why experts in charge of International Standards (e.g. SEFA 9; ANSI AIHA Z9.5: 2003; NFX 15 211) agree to authorize a maximum exposure in the laboratory at 1% of the PEL-TWA for each chemical or one hundred times below the authorized values. Hence, the accumulation of chemical and the “cocktail effect” are drastically minimized and do not represent anymore a health threat for the chemist. A concentration below 1% of the PEL-TWA of each chemical used in the laboratory room can definitely ensure end-users safety.
Fume hood and safety requirements
As demonstrated in the first part, reducing the exposure to chemical is the key factor for laboratory personnel’s safety. Fume hoods have the primary role to lower the chemical concentration possibly inhaled by the chemist. For that reason, it is essential to have a good understanding on how a fume hood works and can provide safety.
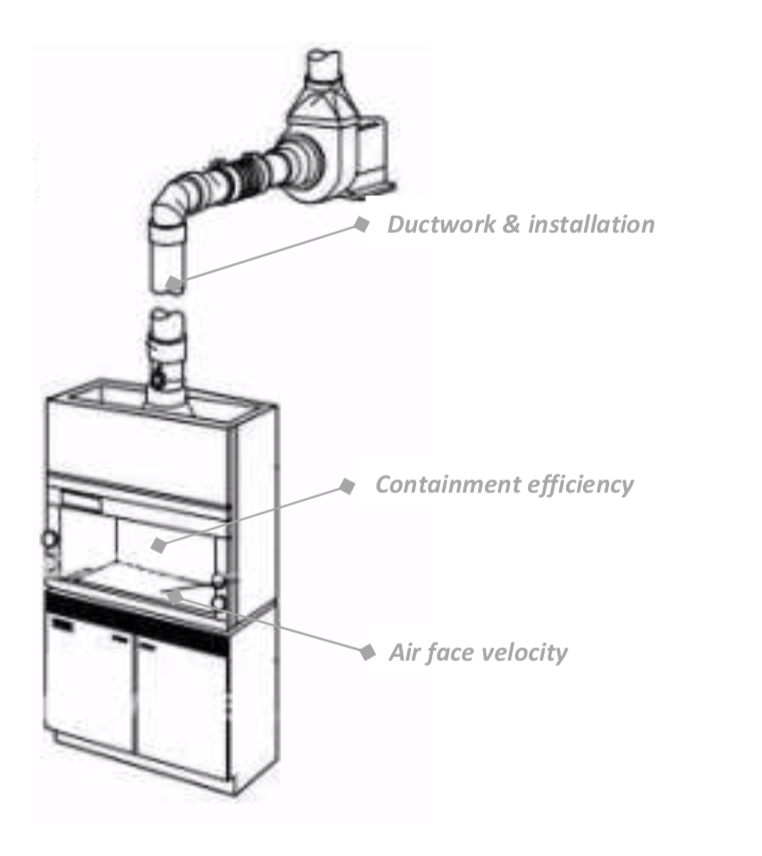
In terms of functions, there are two crucial parts in any type of fume hoods. The first one is the enclosure which plays an essential role to retain any toxic vapors or fumes. As demonstrated by the Graham Law, air molecules collide with the toxic gas molecules and distribute them at a high speed throughout the laboratory air. Toxic gas molecules escaping from a container such as bottle or beaker will travel very quickly in your laboratory: in a fraction of second, a molecule can travel several meters away from the source depending on its molar mass.The enclosure shall play the role of a shield to stop toxic molecules to travel and contain them. But the enclosure is only partially closed with the front sash opening, offering toxic molecule an escape route which could affect the chemist. That’s the reason why the ventilation is also crucial. The ventilation is creating an airflow entering the fume hood at the front sash opening and taking chemical vapors handled inside to outside through the exhaust duct (or through filter in a ductless fume hood).The ventilation of fume hoods has been strictly defined by International Safety Standards (such as in USA with ANSI/ASHRAE 110:2012, ANSI/AIHA Z9.5-2011and in Europe with EN 14175) to prevent harmful exposure of user to hazardous substances. These International Safety Standards have determined two main safety criteria to obtain efficient fume hood ventilation: the air face velocity and the containment efficiency.
The face velocity is related to the speed of air being at the sash plane level and is expressed in meter per second (m/s). According to the International Safety Standards for fume hoods, the air face velocity shall be set between 0.4 and 0.6 m/s on average at the sash opening. It is designed to create an air barrier between the chemical evaporations present in the enclosure and the possible external turbulences around the fume hood, which can eventually provoke Venturi effects and draw air from the inside the hood into the laboratory room through the front sash opening. Such turbulences can be generated from a person walking along the hood, by the air draft of the HVAC system or simply by opening-closing a laboratory door. A face velocity of 0.4 to 0.6 m/s at the sash opening surface has been proven to have the capacity to counter the effects of external turbulences in the laboratory room. Air face velocity can be easily measured by a simple and inexpensive test with a hot wire anemometer.
For a long time, the face velocity set at around 0.5 m/s was the only criteria to consider a fume hood as safe; but in the eighties, researches showed that chemicals could easily escape through the front sash, even with 0.5 m/s face velocity, mainly due to strong air turbulences inside the fume hood enclosure. By 1985, the OSHA in USA introduced in the ASHRAE 110 Standard a second safety criteria called the “containment efficiency” of the fume hood, or the ability of a fume hood to keep the chemicals vapors inside the enclosure and avoid them to escape through the front sash opening. A new test was introduced consisting in emitting a tracer gas, namely Sulfur Hexafluoride (SF6) at 4 liters per minute in the enclosure and verifying at the nose level of a manikin placed in front of the hood that a minimized quantity of SF6 will be detected. To pass the containment test, the concentration measured at the manikin level shall be less than 0.05 ppm of SF6 (ANSI-ASHRAE Z9.5). Although this test is as important – if not more important than the face velocity test – it has been ignored by many fume hood manufacturers since it is more complicated and much more expensive to perform. This test shall be performed to test fume hood samples in ideal conditions in a testing laboratory to verify that the fume hood’s design is appropriate to obtain the 0.05 ppm of SF6, but also after it has been installed at the customer’s site where 0.1 ppm of SF6 is accepted.
Consequently, lab managers should first request the fume hood manufacturers to provide test reports, confirmed by an official independent testing party, for air face velocity and containment efficiency before any purchase. Secondly, they should request the manufacturers to proceed to those tests after the installation of the fume hoods on-site, to verify that they are well installed, with adequate auxiliary air, in a non- turbulent area and can provide full safety to the chemists.
Ductless fume hood, a safety and sustainable alternative solution
Traditional ducted fume hoods are no longer the only solution offered to the laboratory. The development of filtration technology for the past 48 years, now gives laboratory workers more options when choosing a new fume hood without compromising their safety.

Inspired by the activated carbon technology found in modern gas masks, a ductless fume hood is a vented enclosure fitted with filter cartridges designed to retain the chemical vapors handled inside. Ductless fume hood are therefore no more dependent from any external ductwork and auxiliary air supply systems, and can be easily installed anywhere on any existing work bench. Since no ductwork is required, a ductless hood eliminates the direct discharge of pollutants into the atmosphere, fully contributes to the protection of the environment and does not consume any laboratory air-conditioned air, since they recycle the purified air back into the laboratory room.
In terms of safety, ductless fume hoods are submitted to the same requirements as for conventional ducted hood in respect to air face velocity (between 0.4 to 0.6 m/s) and containment efficiency (below 0.05 ppm of SF6 tracer gas). Additionally, it shall also offer full guarantee at the filtration level to avoid any release of chemical into the laboratory room. The filtration efficiency of ductless fume hoods are ruled by International Safety Standards such as the AFNOR NFX 15-211 (French), the SEFA 9 (USA), the JG/T385 (China), the DIN 12927 (Germany). These standards establish specific performance criteria for the quality of the filtered air that a ductless fume hood can return into the lab room.
First, the concentration of a chemical downstream the filter shall not exceed more than 1% of the PEL-TWA of this chemical. Secondly, the ductless fume hood manufacturer shall provide an exhaustive list of common chemical agents that he certifies can be manipulated in its ductless fume hood and indicating the retention capacity for each chemical at a filtration efficiency of 1% of the PEL-TWA of the chemicals Thirdly, a thorough chemical assessment must be conducted by the manufacturer with each user to ensure the filtration efficiency, determine the most suitable filter and evaluate its lifespan, and also to advise the most suitable filter saturation detection system. Lastly, a certificate must be placed at the front of ductless fume hood unit after installation, mentioning the chemicals authorized to be used and the filter life.
Using a ductless fume hood requires discipline from the chemists. They must ensure that any chemical handlings have been validated beforehand by the manufacturer and to follow operations as described in the initial assessment. On the other side, fume hoods featuring filtration technology allow complete flexibility to laboratory workers since they can be installed anywhere in the lab and easily relocated. Ductless fume enclosures require no advance planning and are immediately available for use with almost no installation costs. Mobility is inherent with this design as only an electrical socket and adequate bench top space are required to locate the ductless hood.
Conclusion
Obtaining a good protection of the chemists in a laboratory requires a good understanding of the safety issues with chemical exposure, a good understanding of the safety criteria of fume hoods, a good discipline of the chemists using them. Such a safety approach is not simple: ducted fume hoods are complex to install and require high expertise from the manufacturers to make them working efficiently. Ductless fume hoods are easy to install, offer an excellent containment, but require a high expertise from manufacturers to offer excellent filtration efficiency and high retention capacity. Both systems shall be combined in the laboratory to provide the most adequate safety to the chemists, ducted fume hoods mainly for performing large duties involving large emission of chemicals, while ductless hoods shall be used for small and medium duties, involving well controlled emissions of chemicals.
Courtesy Dominique Laloux,General Manager, Kunshan Erlab DFSL, China Teddy Pitot, Area Sales Manager, Erlab Southeast Asia, Malaysia
